SPORT AND PROTIDES in partnership with ATLET
Recap'
Proteins constitute a heterogeneous chemical family and are considered to be biomolecules of primary importance:
Quantitatively, proteins represent 55 to 85% of dry weight. They are the second most common element in the body after water.
Qualitatively, they have a structural role but also a vital functional role.
Except in exceptional cases (prolonged fasting, insufficient glycogen reserves, etc.), proteins do not contribute significantly to meeting energy needs.
Our needs are very important in terms of proteins. Our organization manufactures nearly 100 different kinds!
All proteins are built from 20 different amino acids.
Among them, 8 are essential amino acids (EAA): the human body does not know how to manufacture them. The food must therefore provide them and what is more, at the same meal. Because, if to produce one of its proteins, the body lacks only one of these 8 EAAs, the production of the protein stops, the body not knowing how to put aside the other 7 until the 8th arrives.
Animal proteins provide all of these 8 EAAs, which is not the case with vegetable proteins, which are deficient in one of the 8 EAAs. Vegetarians must therefore eat at the same meal both cereals (wheat, corn, rice, etc.) low in lysine and pulses (lentils, chickpeas, etc.) low in methionine.
Protein synthesis is essential for development, growth, but also for maintaining body mass. If carbohydrates represent the essential source of energy supply, regular exercise significantly increases the daily requirements of nitrogen compounds. Under very specific conditions, certain amino acids are likely to be oxidized to thus constitute energy substrates in their own right. However, all the proteins present in the body play a specific functional role, and there are no stored and stored amino acids like carbohydrates or lipids. If necessary, it is therefore the amino acids derived from structural or functional proteins that will be used, which is likely to affect the functioning of the organism.
So, in the absence of protein intake, the body self-cannibalizes and self-digests!
The absence of proteins obliges the body to draw them from its “reserves”: the melting of the muscles (including the heart) and then of the viscera (intestinal, liver, etc.) is then inevitable!
Drawing on the reserves leads to deficiencies: the immune defenses weaken, digestion and transit slow down, healing is bad, the skin ages...
As is often the case, the variations recorded in protein metabolism are closely linked to the type of sport practised, and the problems posed will be very different depending on whether the sport discipline considered is endurance or strength-power. However, in the very wide range that goes from short and very high intensity exercises (anaerobic exercise of the explosive type) to long-duration exercises of the enduring type, the responses of protein metabolism are qualitatively similar, associating a drop in protein synthesis and increase in degradation during activity, and the reverse during recovery…
Reminders of biochemistry lessons …
Proteins constitute a heterogeneous chemical family and are considered to be biomolecules of primary importance:
- quantitatively, proteins represent 55 to 85% of the dry weight. They are the second most common element in the body after water.
- qualitatively, they have a structural role but also a vital functional role.
Except in exceptional cases (prolonged fasting, diabetes, etc.), proteins do not contribute significantly to meeting energy needs.
They have a role of mechanical support and tissue support, for example collagen, the most abundant protein in the body; at the cellular level, cytoskeletal proteins (actin, tubulin) are responsible for cell shape.
They have a role of biochemical catalyst, case of enzymes without which almost all chemical reactions would be impossible in the body; role of blood carrier, albumin (which is the most important plasma protein, contributes to the transport of free fatty acids or certain vitamins) or hemoglobin (located in the red blood cells, allows the transport of oxygen and carbon dioxide) ; role of membrane transporter, proteins quantitatively and qualitatively control the exchanges between the cell and the extracellular environment, the specific transporters of glucose; role of chemical mediator like peptide hormones such as insulin and glucagon; role of membrane receptor; role in maintaining the integrity of the body, immunoglobulins (antibodies); role of movement, the contractile proteins of the muscles (actin and myosin).
Proteins are organic compounds made up of carbon (C), hydrogen (H), oxygen (O) and nitrogen (N) to which sulfur (S) is sometimes added. Their monomeric structure is amino acid. Depending on the importance of the polymerization and the composition, it is possible to distinguish several types of proteins:
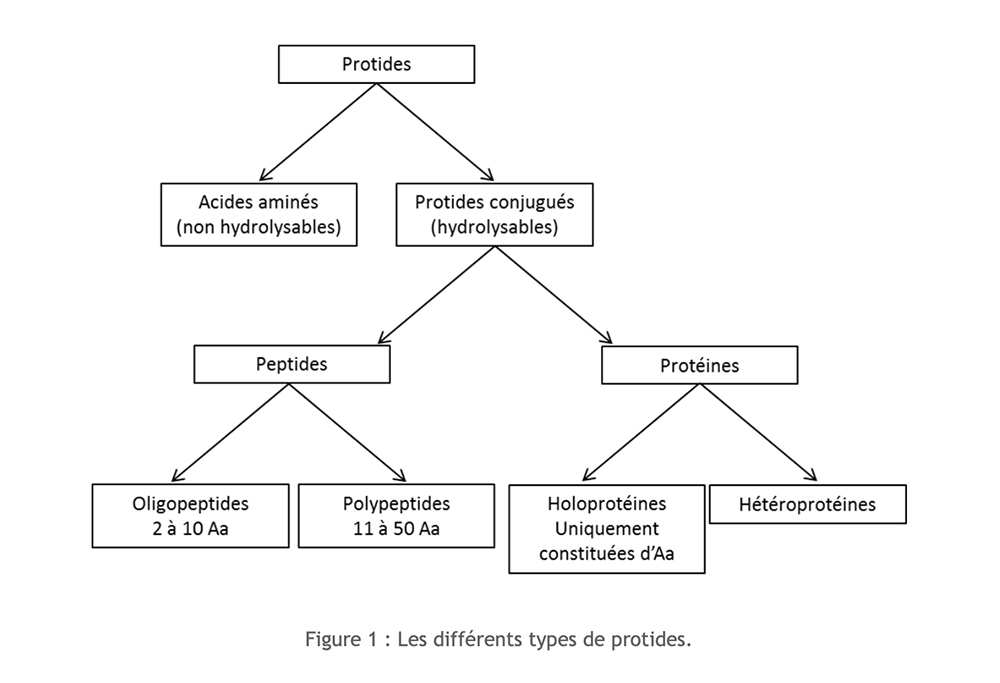
Amino acids
Amino acids have a common molecular structure. They are nitrogen compounds.
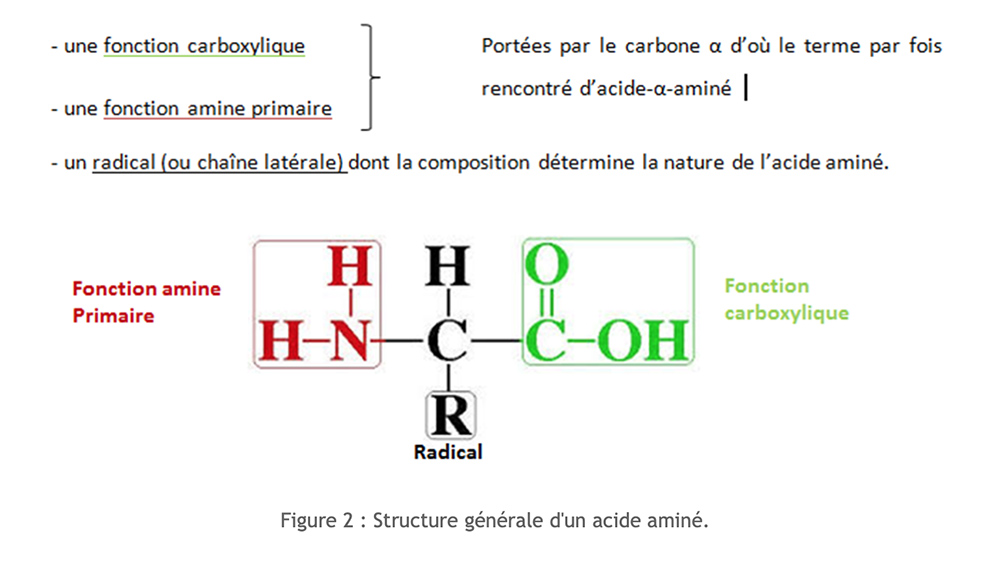
Over 250 different amino acids are listed. However, all of our proteins are made from a group of 20 amino acids, which are calledstandard amino acids.
We can classify these amino acids according to the nature of their side chain. In order to facilitate the writing of the amino acids, a code of three letters or a code with one letter are used.
The different amino acids
We can list eight essential amino acids in adults (Val, Leu, Ile, Thr, Met, Lys, Phe and Trp) plus a ninth in children (His). These amino acids must imperatively be present in the diet.
- wisteria (Gly or G).
- alanine (Ala or A), amino acid very common in proteins.
- the valley (Val or V).
- Leucine (Leu or L) and isoleucine (Ile or I) which cannot be synthesized by the body, they are therefore part of the essential amino acids.
- Serine (Ser or S).
- Threonine (Thr or T), which is an essential amino acid.
- cysteine (Cys or C) contributes to the stabilization of the tertiary structure of proteins thanks to the formation of disulphide bridges. Cysteine is also the precursor of taurine.
- Methionine (Met or M) which is part of the essential amino acids.
- Aspartic acid (Asp or D) and theglutamic acid (Glu or E). These amino acids are very common in proteins. As free amino acids, they play an important role in nitrogen metabolism. Glutamic acid also serves as a precursor for the formation of γ-aminobutyric acid (GABA), a mediator of the central nervous system.
- asparagine (Asn or N) and glutamine (Gln or Q); they have an important role in nitrogen metabolism.
- Lysine (Lys or K) is one of the essential amino acids (it is found in particular in collagen).
- Arginine (Arg or R) plays an important role in the urea cycle and participates in the formation of creatine.
- Histidine (His or H) is considered an essential amino acid in children.
- Phenylalanine (Phe or F) is one of the essential amino acids. As its name suggests, its structure is that of alanine substituted by a phenyl group, forming a hydrophobic radical. Its hydroxylation gives tyrosine (Tyr or Y). These two amino acids are important because they serve as precursors for the biosynthesis of catecholamines (the most common being adrenaline, noradrenaline and dopamine). Tyrosine participates in the formation of thyroid hormones.
- Tryptophan (Trp or W) is essential amino acid. It is a biosynthetic precursor of serotonin and vitamin B3.
- Proline (Pro or P). Like lysine, it has the particularity of being hydroxylated within collagen: hydroxyproline.
The peptides
Peptides result from the association of amino acids. The bond results from the condensation between the carboxylic function (of the α carbon) of an amino acid and the amine function (of the α carbon) of a second amino acid. This condensation is accompanied by the release of a water molecule.
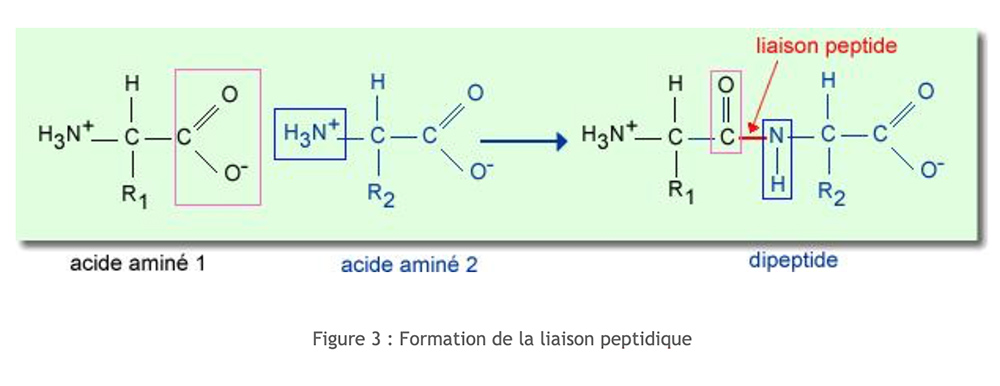
This electronic arrangement induces a rigid and planar peptide bond. As a result, there is no rotation between C and N, which considerably influences the secondary structure of peptides and proteins.
Examples of peptides of biological interest
- Glutathione: This tripeptide plays an important role at the cellular level by neutralizing free radicals, especially in red blood cells. Its sequence is: γGlu-Cys-Gly.
- DHA: This peptide is synthesized by the hypothalamus through its neurosecretory neurons. Released at the level of the posterior pituitary in the blood, it has a hormonal role: it stimulates the reabsorption of water by the kidney.
Protein intake and exercise
Today, it has been scientifically demonstrated that protein metabolism is affected by the practice of exercise. However, we often give them too much of a role; The nutritional reality is more measured.
Protein synthesis is essential for development, growth, but also for maintaining body mass. The regular practice of sport significantly increases the daily needs of nitrogen compounds and in particular in very specific conditions (depletion of glycogen reserves, sharp drop in blood sugar, etc.). However, we do not store amino acids. If necessary, it is therefore the amino acids derived from structural or functional proteins that will be used, which is likely to affect the functioning of the organism.
Endurance sports: Amino acid oxidation and protein requirements
If the need for proteins represents the optimal quantity of proteins necessary to ensure all of the protein synthesis of the organism, to compensate for the oxidation of amino acids and the relative losses due to the acceleration of the turnover of proteins, we can easily conclude that the repetition of endurance exercises induces an increase in the nutritional requirements of proteins and amino acids. Long-term exercise induces significant changes in protein metabolism. Experimental studies show that this type of endurance practice is associated with a drastic reduction in muscle protein synthesis processes. Therefore, in the context of prolonged exercise, if the supply of glucose is insufficient, there is an increase in protein degradation with the aim of increasing the availability of amino acids that can contribute to gluconeogenesis or enter the cycle of Krebs to provide energy in the form of ATP. Finally, the recuperation of this type of exercise will require considering targeted protein intake.
The amino acids available in our body have several origins: they derive from dietary intake, result from endogenous proteolysis, or are synthesized de novo within the body (for non-essential amino acids). The availability of essential amino acids depends only on dietary intake and their level of degradation.
Excluding exercise, there is a perfect balance between degradation (proteolysis) and protein synthesis (proteosynthesis).
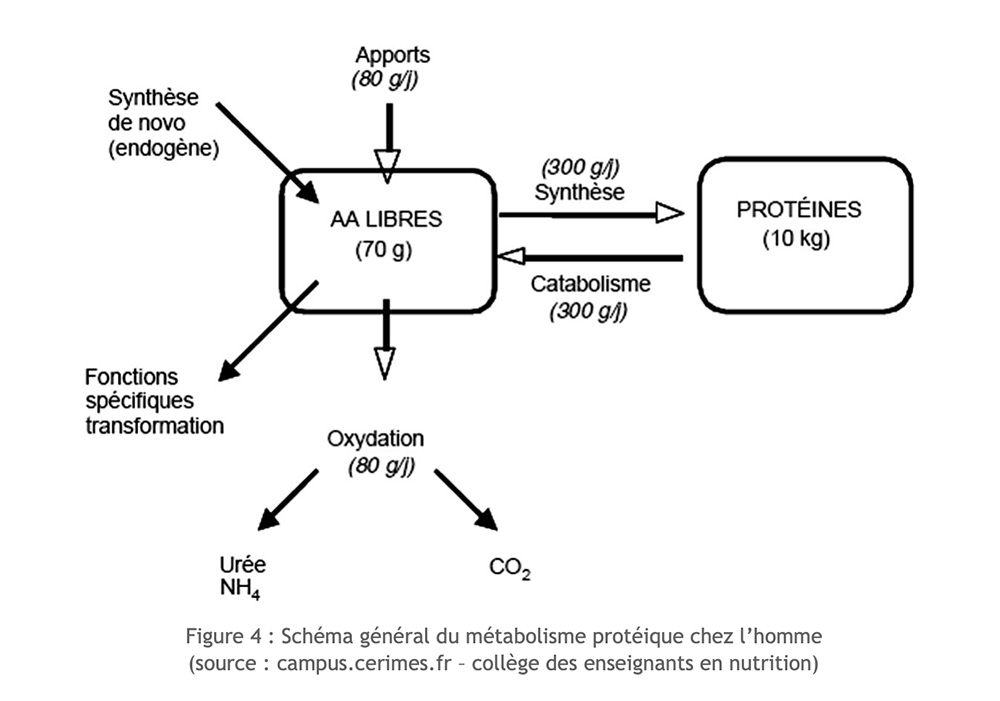
But in the context of long-term exercise, nitrogen intake is reduced, protein synthesis cannot compensate for its degradation and muscle mass atrophies. Certain amino acids can then be considered as real useful substrates for muscular functioning. They are used oxidatively. However, only a few amino acids can be directly oxidized within skeletal muscle: these are essentially branched or branched amino acids (AAB: leucine, isoleucine, valine), and much more incidentally aspartate, asparagine and proline.
The proteins in our diet
Protein from food never enters our tissues directly. It must be “degraded” into amino acids or dipeptides. Once the digestion phase has taken place, the amino acids ingested via our food intake are indistinguishable from those resulting from the degradation of body proteins. To develop its own proteins, our body will draw from the "common bag" where all the available amino acids are found (those from food and those resulting from the destruction of body proteins), at time "t". . In an ideal situation, our ration should provide optimum levels of amino acids, in particular the eight essentials.
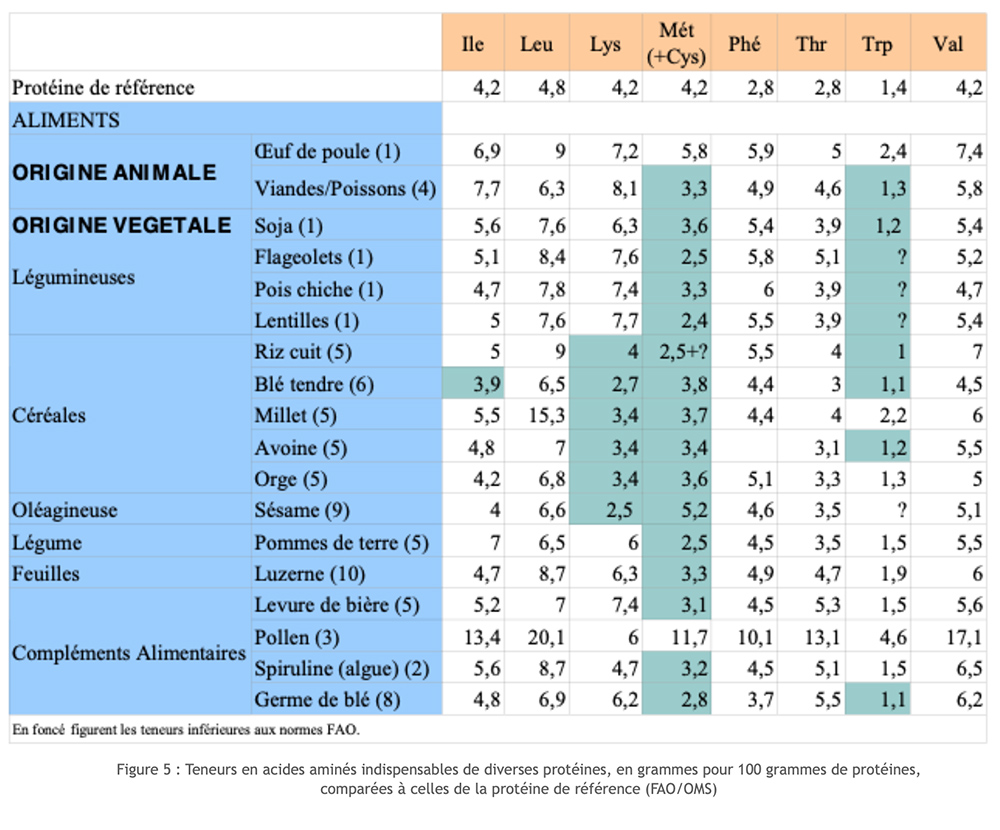
Animal sources of protein score quite well on the Biological Value (BV) and Digestive Utilization Coefficient (DUC) scales. This is different for vegetable proteins. Indeed, whether it is cereals or pulses, we find that some of the essential amino acids are only provided at levels that are far too low (limiting factor). Cereals are generally lacking in lysine; legumes and soya have the limiting factor methionine. By coupling the two sources, we manage to reconstitute a complete set of essential AA, but with a certain waste! Moreover, if this choice makes it possible to prevent severe and chronic deficiencies for an essential AA, it does not guarantee the correct progress of all the metabolisms involving the participation of amino acids over the long term. This functional dissatisfaction is encountered in particular with the syntheses of neurotransmitters, particularly that of serotonin which depends on tryptophan, amino acids that are very often limiting.
Added to this is the problem linked to the amino acid uptake systems. These enter cells through receptor proteins in cell membranes. There are 4 families of receptors for 20 amino acids. In fact, competition phenomena take place. Thus, it is not enough to provide an amino acid at the correct rate to be sure to see the resulting processes take place correctly. Competing amino acids must not be found at too high a rate, which would hinder the assimilation of the first.
The raw amino acid composition of a meal does not allow us to deduce to what extent the supply of different amino acids will be optimally provided to each of our cells.
When you eat protein, you incorporate nitrogen into your body. When we proceed to the difference of the two (input minus output) we obtain what is called the “nitrogen balance”. A positive nitrogen balance is recorded when the nitrogen intake exceeds the sum of urinary, faecal and sweat excretions. A positive nitrogen balance is necessary to ensure an adequate level of synthesis. It is on this method of analysis that the RNP (Nutritional recommendations for the population) are defined, which refer to an essentially quantitative notion of needs, and meet the objectives of preventing deficiencies and not affecting protein synthesis.
The recommendations for endurance sports enthusiasts are consensus: 1,2 to 1,5 g/kg/day.
So, for a person weighing 70 kg, the protein intake recommendation is between: 84g/day to 105g/day; this is equivalent to, for example, very large quantities of meat: from 465 to 580 g! Hence the interest of consuming other sources of protein (cereals, legumes, etc.), or targeted concentrates for early recovery. Once again, we recommend adopting a varied and balanced daily diet, adapted to the level of training.
In athletes, the needs related to functional renewal are increased, under certain conditions, due to the catabolism of proteins, that affecting in particular the contractile elements of the muscle (especially in runners, due to the shock wave which exerts a destructive effect by repeating itself each time the foot presses on the ground). These processes increase the losses and are added to those which correspond to the energetic use of certain amino acids. As a result, a loss of the plasma level of most amino acids and an increase in urinary nitrogen excretion can be observed after a 100 km pedestrian test.
But beware, the increased use of “non-targeted” protein supplements can significantly increase requirements; indeed, the rate of synthesis is fixed by the availability of the least present amino acid in the tissues. This means that if the branched amino acids have seen their level decrease in response to exercise, the syntheses carried out in post effort will be proportional to the remaining quantities of leucine, isoleucine and valine.
Certain amino acids act as molecules capable of stimulating protein synthesis. Leucine is able to specifically stimulate protein synthesis in the muscles and the liver, even under unfavorable conditions (Cf.: Buse MG, Reid M (1975): Leucine, a possible regulator of protein turnover in muscle. J Clin. Invest., 58: 1250).
The intake of protein supplements rich in branched amino acids therefore makes it possible to avoid muscle wasting as well as the deterioration of a certain number of physical, psychological and physiological parameters (Cf.: Degoutte F, Jouanel P & Coll (2006): Food restriction and performance, biochemical, psychological and endocrine changes in judo athletes.Int. J. Sport Med., 27 (1): 9 – 18.).
In addition to branched amino acids, two others seem to us should not be neglected in the athlete's ration:
Methionine (limiting factor for legumes and soy):
The presence of methionine (Met) at an optimal level conditions the progress of a large number of reactions which are also conditioned by the level of energy intake. Indeed, as we have seen above, Met can contribute to supplying energy to the tissues in an emergency situation. This is done via the Krebs cycle at the expense of other Met metabolic intervention. Therefore, any decrease in the dietary intake of Met leads to a slowing down of these different pathways, in particular that of gluconeogenesis, and this, in order to maintain its minimum level in the cells. Furthermore, the transformation of Met into cysteine to promote the formation of glutathione (participating in the anti-radical cascade), is slowed down in favor of the supply of energy, or cannot take place.
Glutamine, Glycine and Aspartate:
Our energy capital is based on the DNA molecule which has the shape of a double helix (a double strand). RNA, on the other hand, is a single-stranded polymer similar to DNA. DNA stores genetic information in the cell, while RNA is used to transmit coding information outside the cell nucleus and then to synthesize proteins from this information. The bases that make up RNA are provided in small quantities by food, the main part actually comes from syntheses carried out in cells. These are made from amino acids, some of which are only available in limited quantities. Genetic capital and proteins are therefore closely dependent. The precursors are: glutamine, glycine and aspartate. Glutamine is all the more involved as it contains 2 nitrogen molecules. Its availability is therefore crucial in situations where rapid cell multiplication is essential (immune response, healing, etc.). Here, the presence of enzymatic cofactors such as zinc, vitamins B9, B12 (from animal sources only) and B6 proves to be decisive for the smooth running of the synthesis process.
Therefore, any deficit or imbalance in the intake of these amino acids, and in particular glutamine, will affect recovery, the response to activity.
These different points make it very complex to define an ideal/optimal protein intake. The recommendation, seen above, takes into account possible functional disorders in order to adapt the catch both quantitatively and qualitatively, on the diversity. The supplements only constitute small readjustments of the ration and consist of highly digestible and targeted peptides. They must not, under any circumstances, compensate for proven nutritional deficiencies. Sometimes considered superfluous, it seems however that they are accompanied by an improvement in the state of health of the athlete concerned.
Article written for our partner Atlet by:
Caroline JOUCLA • State-certified nutritionist-dietician • www.carolinejoucladieteticienne.com
